As chemical compounds are oxidized, they produce free radicals at a cellular leval. Antioxidants react with, and neutralize free radicals. When this happens the destructive properties of the radical is eliminated. A free radical is simply a chemical compound that contains one or more unpaired electrons. One of the ways free radicals are produces is by sun exposure. It does not matter if you lay on the beach for 20 minutes unprotected, or if you walk to work five minutes per day unprotected. Your skin does not know the difference. All sun exposure causes free radicals.
The step that produces a free radical is called the initiation step. Radicals are represented in chemical formulas by a single dot. Below is an illustration of three initiations steps that produce free radicals
Radicals are generally more reactive than non-radicals because they are not paired up. Because they are not paired up, they search out ways of finding a pair for their extra electrion. In their haste to take up a new electron they will attack nearby chemical compounds. Some of these chemical compounds are very important, like your DNA or your skins collegen. If their chemical structure is changed, their function in your skin will also be changed. Once the free radical steals or donates an electron, as second free radical is produced which can react similarly on neighboring molecules. Then, an entire chain reaction has started. As a result, we see skin cancer, destroyed skin elasticity, hyper pigmentation, discoloration and premature wrinkling.
There are a few ways we can stop this from happening to our skin. The first is simply to avoid the sun. Unfortunately we cannot live indoors every day. We must walk outside to our car, go shopping, work and pick up the children. Therefore, broad band SPF sunscreen should be a daily part of your skin routine. There is never...never...never a reason to walk outside without sunscreen. I personally never go outside without sun protection. I have at least three types of sunscreen that I use every single day, rain or shine.
However, even if you are as vigilent as I am when it comes to protecting your skin from sun damage, some sun light will manage to get past the sunscreen or through your clothing. This is where your knowledge of anti-oxidants comes into play. Cell membranes contain a chemical called alpha-tocopherol. Alpha-tocopheral picks up free radicals, transport electrons, and neutralizes free radicals.
Topical anti-oxidants are classified into two broad divisions depending on whether they are soluble in water or fats. Water soluble antioxidants react in the cell fluids and blood plasma. Lipid-soluble antioxidants protect cell membranes. The amount of protection provided by an anti-oxidant will also depend on its concentration.
Ascorbic Acid ( water) Vitamin C
Glutathione ( water)
Lipoic Acid ( water)
Uric Acid ( water)
Carotenes (fats) Also known as Beta-carotene or Vitamin A
Tocopherol ( vitamin E ) (fats) Vegetable Oils
Ubiquinol ( coenzyme Q) (fats)
Selenium ( Brazil Nuts)
coenzyme Q10 Oil soluble vitimin like substance. Soybean Oil,. Olive Oil, Grapeseed Oil, Avocado
Apply topical antioxidants only at night. Any sunlight will neutralize the effects of antioxidants. Make sure that they are stored in light proof containers. If topical antioxidants are stored in clear containers they will become ineffective.





 10:31 AM
10:31 AM
 Rue
Rue

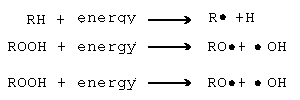









0 comments:
Post a Comment